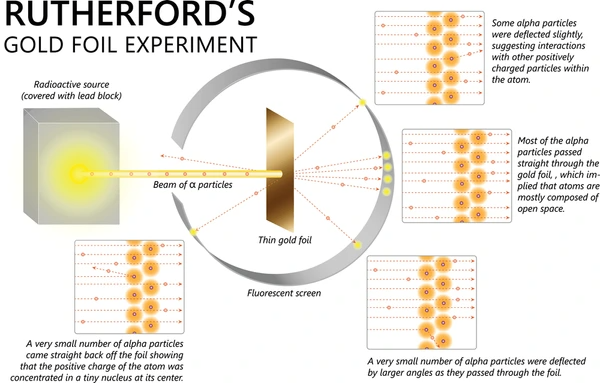
The Gold Foil Experiment, conducted by Ernest Rutherford in 1909, was a monumental experiment that transformed our understanding of atomic structure. It provided the first evidence of the existence of a dense, positively charged nucleus at the center of an atom, fundamentally changing the field of chemistry and physics.
Historical Context
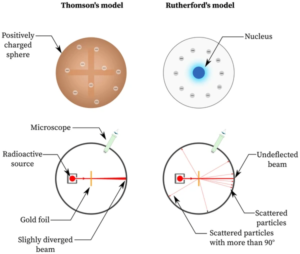 >
>- J.J. Thomson’s Plum Pudding Model (1904): Prior to Rutherford’s experiment, the prevailing model of the atom was proposed by J.J. Thomson. In this model, the atom was thought to consist of a sphere of diffuse positive charge, with negatively charged electrons embedded within it like “plums in a pudding.”
- Challenge: Rutherford set out to test this model by examining how alpha particles (helium nuclei) interacted with a thin sheet of gold foil. The experiment was designed to investigate the distribution of positive charge within the atom.
Experimental Setup
- Alpha Particle Source: A radioactive substance emitted high-energy alpha particles, which are positively charged and relatively heavy.
- Gold Foil: A very thin sheet of gold, only a few atoms thick, was used as the target for the alpha particles.
- Detection Screen: A circular zinc sulfide screen surrounded the gold foil. When an alpha particle struck the screen, it produced a tiny flash of light, allowing the direction and angle of deflection to be recorded.
- Vacuum Chamber: The experiment was conducted in a vacuum to prevent interference from air molecules.
Expected Results (Based on the Plum Pudding Model)
- According to Thomson’s model, the positive charge in the atom was spread out uniformly. As a result, Rutherford and his team expected that the alpha particles would pass through the gold foil with very little deflection. They anticipated that almost all the alpha particles would pass straight through, with perhaps a few experiencing small angles of deflection.
Observed Results
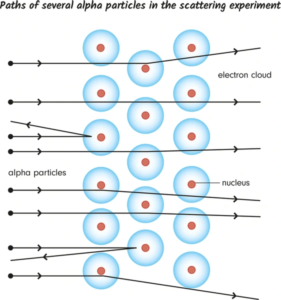 >
>- Most Particles Passed Through: As expected, the vast majority of alpha particles passed through the gold foil without any significant deflection.
- Some Particles Deflected at Large Angles: To the astonishment of Rutherford and his team, a small percentage of the alpha particles were deflected at large angles, with some even bouncing straight back towards the source.
Rutherford famously remarked: “It was as if you fired a 15-inch shell at a piece of tissue paper, and it came back and hit you!”
Rutherford’s Conclusions
The unexpected large deflections could not be explained by the plum pudding model. Rutherford proposed a new model of the atom based on the following conclusions:
- Existence of a Nucleus: The large deflections indicated that the positive charge of the atom was not spread out, as Thomson suggested, but rather concentrated in a tiny, dense core, which Rutherford called the nucleus. This nucleus contained nearly all of the atom’s mass and all of its positive charge.
- Atoms are Mostly Empty Space: Since most alpha particles passed through the foil with little or no deflection, Rutherford concluded that the atom was mostly empty space, with the electrons orbiting the nucleus at relatively large distances.
- Nuclear Model of the Atom: In this new model, the atom consists of a dense central nucleus surrounded by a cloud of electrons. The nucleus is tiny compared to the overall size of the atom, which explains why most alpha particles passed through the foil without deflection.
Significance of the Experiment
- End of the Plum Pudding Model: Rutherford’s findings disproved the plum pudding model of the atom and introduced the nuclear model, which forms the foundation of modern atomic theory.
- Birth of Nuclear Physics: The discovery of the nucleus led to further investigations into the structure of the atom, including the identification of protons and neutrons, and eventually, the development of quantum mechanics.
- Influence on Future Research: Rutherford’s model laid the groundwork for Niels Bohr’s model of the atom, which introduced quantized electron orbits, and later, for the development of quantum mechanics and our modern understanding of atomic structure.
Key Points to Remember
- Alpha Particles: Positively charged particles emitted by radioactive decay.
- Gold Foil: Extremely thin sheet of gold used to study the interaction of alpha particles with matter.
- Nucleus: A dense, positively charged core at the center of the atom, containing most of its mass.
- Electrons: Negatively charged particles orbiting the nucleus at relatively large distances.
- Atomic Structure: The atom is mostly empty space, with a central nucleus surrounded by electrons.
Diagram of the Experiment
 >
>- Alpha Particle Source: A radioactive source emitting alpha particles.
- Gold Foil: The thin gold foil target.
- Detection Screen: A circular zinc sulfide screen that detects deflected alpha particles.
The diagram would show alpha particles passing through the foil, some deflecting at small angles, and a few deflecting at large angles, indicating the presence of the nucleus.
Impact on Modern Science
Rutherford’s gold foil experiment was a watershed moment in the understanding of atomic structure. It led to the realization that the atom is not a uniform sphere but instead has a small, dense core, which fundamentally changed the course of scientific research in both chemistry and physics.